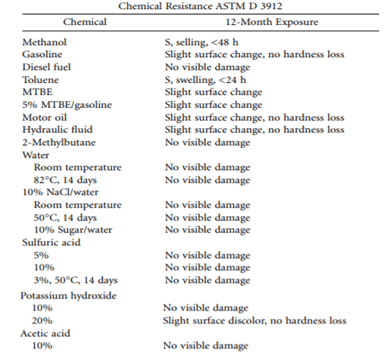
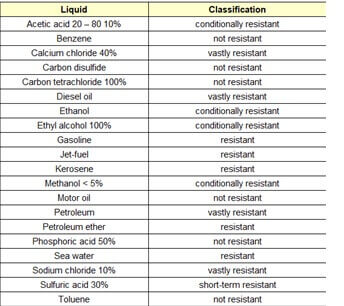
An epoxy primer is required for many polyureas as their adhesion may not be sufficient to withstand heavy-duty service. Because of their short cure time, they are often used with special equipment such as plural component spraying using a spray gun with a mixing head. However, all guidance that is provided by the manufacturer regarding the mixing of multi-component products, thinning requirements, induction times, and special application requirements should be followed.
These aromatic polyurethane floors are suitable for many different levels of chemical and mechanical exposure. They can also be used on concrete, plywood, metal, and wood surfaces. A waterproof membrane can be used in certain cases to extend the application. This coating type can be applied in thick film for immersion service; some formulations allow up to 500 microns (20 mils) or more per coat for industrial and marine applications.
Aliphatic polyurethane coatings (high-performance two-component UV-stable topcoats) are typically used for exterior coatings. However, some formulations are suitable for exposure in environmental zones 1A (interior, normally dry), including offshore structures, chemical and petrochemical plants, bridges, pulp and paper mills, and the power industry, excluding immersion zones.
Polyurea coatings perform highly in industrial applications requiring high abrasion, chemical, and corrosion resistance. They can be used on concrete, metal, wood, ceramic, and other materials for waterproofing and protection. (To learn more about polyurea coatings, read The History and Industry Adoption of Polyurea Coating Systems.)
Different raw materials in chemical reactions and the use of catalysts to accelerate the reaction time can cause marked differences in the sensitivity and environmental conditions that affect the application and cure times of the products. Other physical and chemical characteristics can also be used to indicate different properties.